The more that is learned about how microfluidic processes control or contribute to cellular change, the sooner science will be able to design a cost-effective medical treatment based on that information. New research on microtubules and blood-vessel organoids augments this.
Recent findings by a team of engineering and medical scientists at Stanford University shed new light on how cell components move around and self-renew. Part of the study’s focus was on the link between microtubules and self-organization.
Microtubules, hollow tubes about 25 nm in diameter, are critical for maintaining cell shape and movements. The study analyzed what role microtubules have in maintenance, and found that microtubules are continuously losing and gaining molecules. Understanding the mechanics of how microtubules move around and contribute to regeneration helps to light affordable paths to wound healing in humans and animals.
Cells are known to self-organize at the direction of particular protein structures, via recognized regulators of the organization. However, the Stanford team observed that some cells self-organized in the absence of their known regulators. An interaction between microtubules and other molecules on the membrane surface was the reason.
This observation raised questions about what role microtubules play in the regeneration when other elements of the system are paralyzed. The researchers described how microtubules seemed to be involved in some minimal requirements for cellular self-organization. Identifying this process opens the door to greater understanding of cell renewal.
Clarifying the precise functions of microfluidics has paved the way to viable organoids, on which assessments can be done without having to conduct tests on an entire organism. For example, the University of British Columbia researchers recently were able to architect human vascular organoids that were nurtured to duplicate diabetic blood vessels, and can be used as test models.
The UBC scientists were able to grow human blood vessels as organoids in a lab, for the first time. This spawns investigation of treatments for vascular diseases by highlighting how changes to blood vessels occur. Such changes are a major cause of death among diabetics.

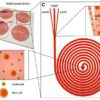
An illustration of vascular organoids, lab-made human blood vessels, based on original data. Credit: IMBA
“Being able to build human blood vessels as organoids from stem cells is a game changer,” said the study’s senior author Josef Penninger. “Every single organ in our body is linked with the circulatory system. This could potentially allow researchers to unravel the causes and treatments for a variety of vascular diseases, from Alzheimer’s disease, cardiovascular diseases, wound healing problems, stroke, cancer and, of course, diabetes.”
Many diabetic symptoms are the result of changes in blood vessels that result in impaired oxygen supply of tissues, and impaired circulation. Not a lot has been known about vascular changes arising from diabetes. This creation of human blood-vessel organoids is a significant step toward tipping the scales from unknowns to knowns.
Determining how to cultivate three-dimensional human blood-vessel organoids in a lab dish is indeed a huge step toward unveiling blood-vessel change mechanisms.
These “vascular organoids” when transplanted into mice, developed into functional human blood vessels, including capillaries and arteries. So not only was it possible to engineer blood-vessel organoids from human stem cells in a dish, but they also grew a functional human vascular system in another species.
The organoids resemble human capillaries to a great extent, even on a molecular level, and can be used to study blood vessel diseases directly on human tissue. The researchers described how organoids can be used to study the lack of oxygen and nutrient delivery to blood vessels that occurs in diabetic patients, causing complications including kidney failure, heart attacks, strokes, blindness, and the peripheral artery disease that leads to amputations.
They were surprised to find the vascular organoids showed expansion of the basement membrane, which is exactly what causes the oxygen-and-nutrient depletion in humans. The damage to the vascular organoids precisely mirrored what is seen in diabetic patients.
Using the vascular organoids for testing, they found that no currently-prescribed anti-diabetic medications had positive effects on these blood vessel defects. But they did find an enzyme inhibitor that prevented thickening of the basement membrane.
The researchers noted that the findings could allow them to identify underlying causes of vascular disease, and to potentially develop and test new treatments for the world’s estimated 420 million people with diabetes.
That many patients should theoretically translate into significant Research and Development endeavors.
Enjoyed this article? Don’t forget to share.

Kathy Jean Schultz
Kathy Jean Schultz is a freelance medical science writer who focuses on medical innovations. She earned a Master’s Degree in Research Methodology from Hofstra University, and a Master’s Degree in Psychology from Long Island University. She is a member of the National Association of Science Writers, and the Association of Health Care Journalists. Her articles about organoids include "Would you trust a 3-D printed mini organ to test your drugs?" and "Stem cells not only slow disease, they come with their own safety test".