Microfluidics advances appear in sometimes surprising ways. One of the most intriguing is their emerging role in microbiome research. A new study shows how some bacteria in the microbiome, inside the human intestine, generate electricity outside of their own cell walls. The potential for microfluidics’ role in this research is significant, considering that the electricity generated in gut bacteria might be harnessed on a large scale — for example, at a bacteria-laden waste processing facility. Microfluidics advances will likely define how the mechanics would work, and therefore how related processes might be useful to bioeconomics, and also to urgent energy-creation challenges.
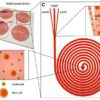
A team of scientists from various institutions, including the U.S. Department of Energy, investigated the mechanics: how do these bacteria transport electrons across cell walls? They probed how Listeria bacteria residing in the human intestine transport electrons — in the form of tiny currents — through their cell wall.
The findings were just one example of bacteria in the human gut that regularly produce electricity. It seems that hundreds of other bacteria may be employing similar, but so-far-unidentified processes. A wide range of bacteria types might be conducting a similar transport of electrons outside their cell wall. But findings suggested that Listeria appeared to use a method that is different from other electricity-producing bacteria.
Does this mean these bacteria could predictably produce electricity outside of their normal environment? The researchers noted that these findings point toward the potential creation of batteries from microbes.
The traditional medical focus has been understanding how bacteria infect — or maintain — a healthy gut. But the findings about electrons have spawned new questions: Is it possible that microbe-derived batteries could generate electricity in large waste treatment plants? Could batteries built from microbes feasibly generate electricity on a grand scale?
“While scientists have found bacteria that produce electricity in exotic environments like mines and the bottoms of lakes, researchers have missed a source closer to home: the human gut,” the researchers noted.
Recent advances outline the potential for microfluidic engineering of microbiomes’ microbial interactions. Microfluidic chips have the capacity to fuel the automated assembly and analysis of microbial communities.
In addition, novel tools provide a framework to evaluate how metabolic networks drive biological processes. There are new platforms for designing microbiomes with specific properties, and to describe what principles govern their interactions. A goal is identifying processes to be manipulated and monitored, including the prediction of metabolic flux throughout interacting networks.
Mapping gut bacteria types in the human microbiome remains incomplete, due to its wide range of bacterial diversity. Technical roadblocks have included figuring out how to isolate single-bacteria. But a new microfluidic-emulsion device has created microdroplets to capture a single bacterium. Single-bacteria isolation could reveal which properties yield the most insight about infection in humans. Additional studies have described microfluidic approaches to creating realistic in-vitro models of the human intestinal microbiome.
It’s been known for a long time that the body produces electricity. Electrocardiograms measure the heart’s electricity. Electroencephalography measures electrical activity in the brain. Brushing of very dry human hair generates static electricity — with results ranging from being a nuisance, to pain. But the idea that it might be possible to capture enough body-generated electricity for outside-the-body uses, is new.
The human microbiome contains communities of microbes, bacteria, viruses and fungi that function together. Animals have microbiomes controlling their health or sickness too. Soil, water and rocks also contain microbiomes that mediate chemical changes.
Within a microbiome, microbes move around and fuel each other in complicated ways. How we eat, live and use medicine impacts how microbes will interact with each other — working together to maintain health, or to cause disease. Some human microbes cause sickness, while others sustain life.
What’s next for the concept of microbe-produced batteries? Investors are more often mulling partnerships for cutting-edge innovations, from Artificial Intelligence to genetics. A recent Pharmacy and Therapeutics Journal report, “The Human Microbiome Market,” confirms that 260 microbiome therapeutics are currently being evaluated in different stages of development. “The microbiome-based medical products market can be expected to witness substantial growth over the coming decade,” according to the report. “The concept of microbiome-based therapeutics has generated significant enthusiasm within the medical science community, defining a new frontier in the field of medicine.”
A new frontier indeed. These particular findings address two compelling issues: medical advances, and also urgent energy-production problems.
This unique pairing would seem to check a lot of boxes for investors.
Enjoyed this article? Don’t forget to share.

Kathy Jean Schultz
Kathy Jean Schultz is a freelance medical science writer who focuses on medical innovations. She earned a Master’s Degree in Research Methodology from Hofstra University, and a Master’s Degree in Psychology from Long Island University. She is a member of the National Association of Science Writers, and the Association of Health Care Journalists. Her articles about organoids include "Would you trust a 3-D printed mini organ to test your drugs?" and "Stem cells not only slow disease, they come with their own safety test".