Although scaling up for mass production remains an ongoing challenge in the world of microfluidics, other developments are driving positive predictions. For example, the microfluidic immunoassay market recently welcomed a report by India-based Meticulous Research that includes international growth estimates of more than $2 billion by 2025. National University of Singapore researchers have developed a microfluidic chip to test for the presence of cancer that would cost only about $50 per test. And Pennsylvania State University biochemist Dr. Paul Cremer just received a $75,000 grant to be used for commercialization of his project, “A Temperature Gradient Microfluidics Instrument for Screening the Colloidal Stability of Therapeutic Protein Formulations.”
Financial potential is being matched medical potential, as microenvironments play a role in new findings about stem cell treatment that may speed patients’ recovery from chemotherapy and radiation. And microfluidics advances have also led to a new tool for diabetes research.
Bone marrow stem cells produce the body’s blood and immune cells, but chemotherapy and radiation suppress that production, and it can take weeks or months for the blood and immune systems to recover. Results of a recent study by UCLA Broad Stem Cell Research Center scientists showed how a newly developed drug compound might hasten the blood system’s recovery after radiation and chemotherapy in mice. Some of the heretofore unknown capacity of microenvironments were critical to this type of investigation.
“The potency of this compound in animal models was very high,” said study co-author Dr. John Chute. “It accelerated the recovery of blood stem cells, white blood cells and other components of the blood system necessary for survival. If found to be safe in humans, it could lessen infections and allow people to be discharged from the hospital earlier.”
Looking at both mouse — and human stem cells (in dishes) — growth factors were observed to promote the recruitment of stem cells from degraded bone marrow, which fueled proliferation and reconstitution. “We’re very excited about the potential medical applications of these findings,” said Chute.
The new compound enabled the blood system to recover sooner from cancer treatments in mice because it was able to stop the molecular process that slows blood stem cell regeneration. “The new compound lifts molecular ‘brakes’ that normally slow the regeneration of blood stem cells,” according to the team.
The new compound speeded up the regeneration of both mouse and human blood stem cells after exposure to radiation. The bloodstream’s own regenerative capacity seemed to power up healing. Angiogenesis, the formation of new blood cells, is controlled by chemical signals, yet many of the mechanisms by which blood cell regeneration uses bone marrow have been unknown. The study highlights the process by which growth factors recruit stem cells from the bone marrow microenvironment. Advances detailing bone marrow’s microenvironments are a significant contribution.
Microfluidic devices can be monitored and imaged using fluorescent markers for tracking, and microenvironments needed to be precisely controlled for this investigation.
Among the mice that received high doses of radiation, almost all that were given the compound survived; more than half of those that did not receive the compound died. Mice that received chemotherapy but no compound had low levels of white blood cells and neutrophils — which fight bacteria — after two weeks; in mice that were treated, white blood cell counts recovered to normal levels within two weeks. The researchers are now refining the process, in preparation for human trials.
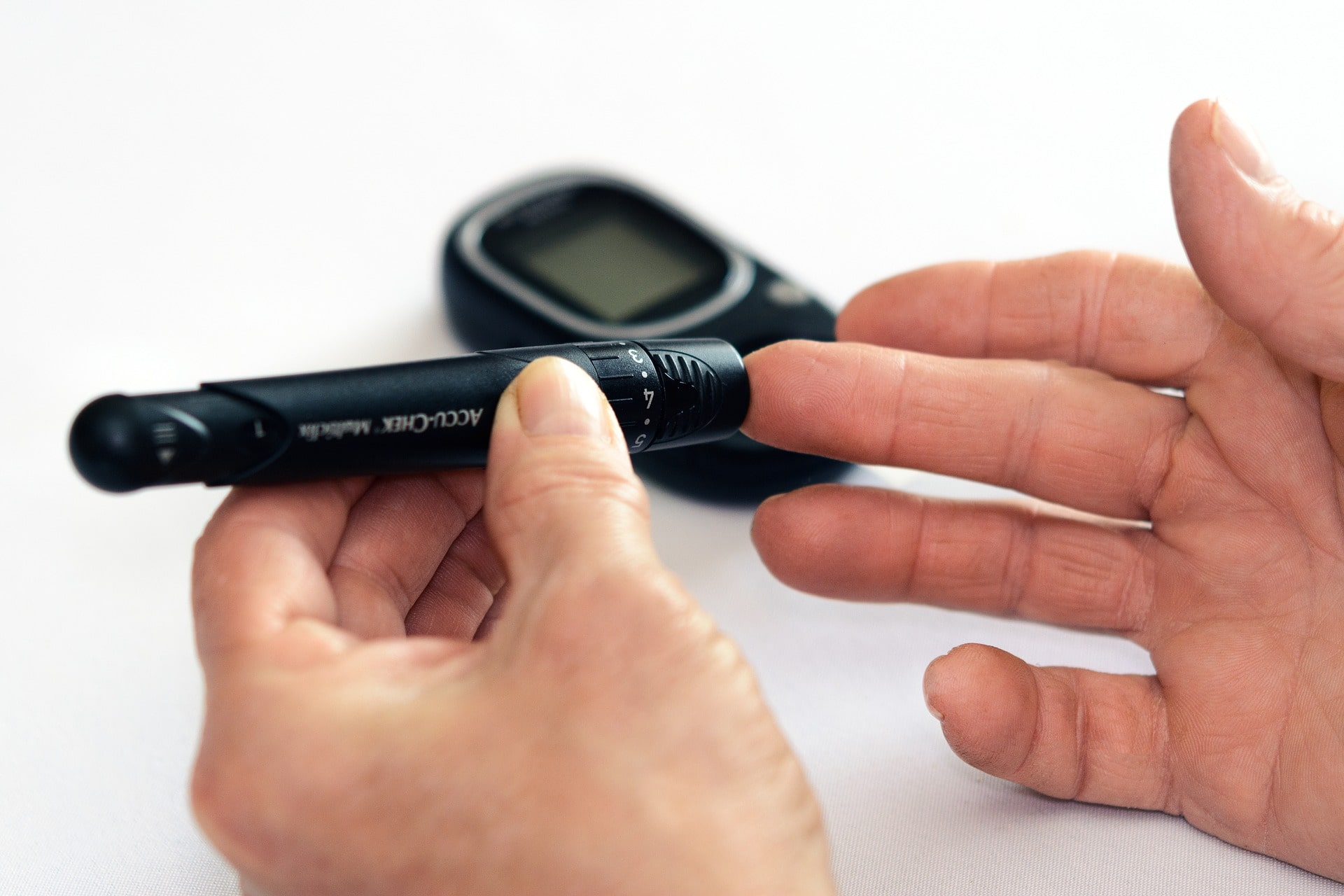
Microfluidics research also has another new arrow in its quiver, in the form of a new tool for diabetes research, developed by a team based at the Harvard Stem Cell Institute. The device improves aspects of studying diabetes, including upgrading the screening process prior to transplantation of insulin-producing cells into a patient.
The team noted that although microfluidic devices have been used to address research limitations in the past, previous adoption has been hampered by “incompatibility of most device materials with large-scale manufacturing. We designed and built a thermoplastic, microfluidic-based Islet-on-a-Chip compatible with commercial fabrication methods, that automates islet loading, stimulation, and insulin sensing.”
The design of their “Islet-on-a-Chip” is based on the human pancreas, where islets process non-stop, incoming information about glucose levels, and adjust insulin production accordingly. The new, automated, miniature device gives results in real-time, which can speed up clinical decision-making.
In addition to diabetes research, the device may prove useful in other areas. Its core technology can be modified to sense an array of microfluidic systems. Because it can detect cell secretions continuously, it can be employed to investigate how cells communicate using protein signals, throughout the body.
“It was exciting to see our lab’s method for measuring islet function taken forward from individual cells to much bigger groups of cells, and incorporated into a device that can be used widely in the community,” said co-author Dr. Michael Roper. “Now, we have a device that integrates glucose delivery, islet positioning and capture, reagent mixing, and insulin detection, and requires far fewer reagents. So labs can use it to do more experiments at the same cost, using a much shorter and easier process.”
Scalable, easier, shorter, and cost-contained. That says it all.
Enjoyed this article? Don’t forget to share.

Kathy Jean Schultz
Kathy Jean Schultz is a freelance medical science writer who focuses on medical innovations. She earned a Master’s Degree in Research Methodology from Hofstra University, and a Master’s Degree in Psychology from Long Island University. She is a member of the National Association of Science Writers, and the Association of Health Care Journalists. Her articles about organoids include "Would you trust a 3-D printed mini organ to test your drugs?" and "Stem cells not only slow disease, they come with their own safety test".