Essential microfluidic techniques for droplet manipulation
In many droplet microfluidics experiments, generating uniform and stable droplets is not enough in and of itself. The droplets normally require a post-generation modification. During the past decade, droplet microfluidics has reached maturity. The technology has evolved to incorporate a variety of techniques for on-chip manipulation of droplets after generation. Mirofluidics manipulation methods are broad and involve approaches for detecting the droplets, merging, splitting, incubating, sorting, and mixing.
Detection techniques for droplet microfluidic
Detection is the inseparable part of manys assay used in microfluidic devices. The mono-disperse droplets that were generated now need to be identified for further processing. The rate of droplet generation is in the order of kHz in many droplet generators. Therefore, the data acquisition and detection techniques for droplet chips need to be operable at high frequencies to be compatible with the ultra-high generation rate of droplets.
Bright-field microscopy is the simplest visualization tool in droplet microfluidics research. It is suitable for analyzing the shape, size, and colour of the droplets. Due to the extremely high production rate of the droplets in microfluidic chips, bright-field microscopes normally connect to a high-speed camera for real-time analysis of the droplets. Bright-field microscopy has been employed for a wide range of applications to date including mechanistic behaviour of the droplets such as merging, splitting, and sorting, mixing, colorimetric study of catalysts, protein crystallization, etc. However, bright-field microscopy has its own shortcomings. It is not practical in cases where a specific compound at low concentrations needs to be monitored. Also, it comes short in the comparative temporal screening of a compound in a single droplet or comparative study of a group of droplets (for e.g. comparison of expression levels of a specific protein within a pool of droplets).
Fluorescent microscopy has the benefits of bright-field with the added advantage of high-sensitivity detection capacity. Fluorescent biomarkers are very common in droplet microfluidics. The marker lies within the droplet and upon activation fluoresce. This gives it a binary nature where the droplets with the desired analytes are on and the rest are off. Also, quantitative and comparative studies are feasible by comparing the fluorescent intensity of the droplets. Fluorescent microscopy is now quite mature in enzymatic activity measurements.
More advanced imaging systems such as laser-induced fluorescence (LIF) and Raman spectroscopy are on the rise for droplet microfluidics experiments. Although imaging systems are quite commonplace in droplet microfluidics assays, the detection methods are not limited to them. Other approaches such as electrochemical detection, capillary electrophoresis, mass spectrometry, nuclear magnetic resonance spectroscopy, and chemiluminescence detection have been integrated with the fabricated microfluidic chips and employed according to the specifications of the experiments.
Picoinjection
 As the name implies, it refers to injecting a tiny amount of a chemical (in the order of pL or less) to the droplets of interest. Picoinjection is of utmost importance after droplet formation. On many occasions, different reagents need to be added to the droplet (microreactor) at different times. Picoinjectors assure that these reagents are added at the right time. Adding a reagent to the droplet can be challenging since the surfactant forms a protective later around the droplet intercepting the reagent. A trigger is needed to loosen this protective layer temporarily to allow the reagent to enter. A wide-spread method for picoinjection is via a pair of electrodes. The electrodes are placed across the injection channel and apply an electric voltage to the droplets that are passing by. This electric voltage ruptures the surfactant layer to let the reagent in.
As the name implies, it refers to injecting a tiny amount of a chemical (in the order of pL or less) to the droplets of interest. Picoinjection is of utmost importance after droplet formation. On many occasions, different reagents need to be added to the droplet (microreactor) at different times. Picoinjectors assure that these reagents are added at the right time. Adding a reagent to the droplet can be challenging since the surfactant forms a protective later around the droplet intercepting the reagent. A trigger is needed to loosen this protective layer temporarily to allow the reagent to enter. A wide-spread method for picoinjection is via a pair of electrodes. The electrodes are placed across the injection channel and apply an electric voltage to the droplets that are passing by. This electric voltage ruptures the surfactant layer to let the reagent in.
Incubation
 Incubation is the bread and butter of droplet microfluidics. It is needed for most of the biological applications of droplet microfluidics. Depending on the duration of the incubation different approaches can be taken.
Incubation is the bread and butter of droplet microfluidics. It is needed for most of the biological applications of droplet microfluidics. Depending on the duration of the incubation different approaches can be taken.
Delay lines are long microfluidic channels where a droplet travels through while being incubated. Delay lines allow continuous droplet formation and incubation. In other words, in delay lines, there is no need to collect the droplets and store them in incubators thus there is no delay between formation and incubation. Delay lines are practical in cases where the incubation time is not too much. Longer incubation time demands longer delay lines. As the delay line becomes larger the pressure drop increases dramatically (use this microfluidic pressure drop calculator) and the amount of reagents needed for the experiment grows. To overcome this, the delay line can be widened. Widening the delay line while keeping the flow rate constant, drops the droplet velocity which translates to a longer delay in the delay line as well as a reduction in pressure drop.
Another approach for incubation can be collecting the droplets and incubating them inside Eppendorf tubes or huge microchambers. After incubation, the droplets can be reinjected to another microfluidics chip for further analysis.
Reinjection
 Reinjection is another popular add-on for droplet microfluidics. Reinjection is needed when the droplets need to be introduced to another chip for post-processing. It is mostly used when droplets need to be incubated followed by reinjection to a droplet sorter chip. The keypoint for reinjection is that the droplets need to enter the second chip in a uniform fashion. Random reinjection of the droplets can disrupt the functionality of the microfluidics chip. For a uniform reinjection, the droplet solution needs to have the minimum amount of the continuous phase possible. By discarding the continuous phase, the droplets will enter the chip in a compact and uniform manner.
Reinjection is another popular add-on for droplet microfluidics. Reinjection is needed when the droplets need to be introduced to another chip for post-processing. It is mostly used when droplets need to be incubated followed by reinjection to a droplet sorter chip. The keypoint for reinjection is that the droplets need to enter the second chip in a uniform fashion. Random reinjection of the droplets can disrupt the functionality of the microfluidics chip. For a uniform reinjection, the droplet solution needs to have the minimum amount of the continuous phase possible. By discarding the continuous phase, the droplets will enter the chip in a compact and uniform manner.
Sorting
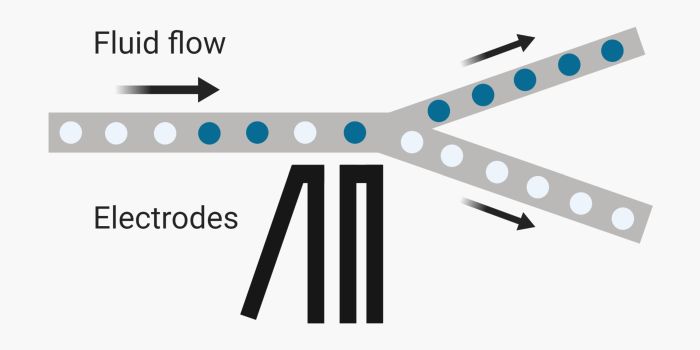 Sorting refers to the phenomenon in which the droplets are sorted based on a particular specification. Oftentimes, this feature is the fluorescence intensity of the droplets. The microfluidics sorter chip recognizes the droplets in which the target marker is active thus fluoresces. Next, the downstream electrodes trigger and push the target droplet to the collection channel. Empty droplets, however, do not follow and go to the waste channel.
Sorting refers to the phenomenon in which the droplets are sorted based on a particular specification. Oftentimes, this feature is the fluorescence intensity of the droplets. The microfluidics sorter chip recognizes the droplets in which the target marker is active thus fluoresces. Next, the downstream electrodes trigger and push the target droplet to the collection channel. Empty droplets, however, do not follow and go to the waste channel.
The microfluidics droplet sorters have to be quick in both detection and actuation to comply with the ultra-high droplet generation rates. These sorters often operate at ~kHz ranges. Mechanical, electrical and magnetic actuators have been used for droplet sorting so far. In general, they all follow the same principle. The chip consists of a droplet detection section, a bifurcation, an actuator close to the bifurcation, a waste channel, and a collection channel. Normally, one of the microchannels has a slightly lower hydrodynamic resistance. Therefore, the droplets naturally flow through this channel when the sorter is off. In the on-mode, a data acquisition system detects the droplets, sends a signal to the actuator, and the actuator causes the droplets to move to the other channel. Depending on the actuator type, this part can be different. Some of the sorters deflect the waste channel such that its hydrodynamic resistance increases thus the target droplet moves to the collection channel instead. Other methods focus on changing the path of the droplet rather than deflecting the channels. The actuator exerts a force on the droplet upon activation forcing it to change its natural path and move to the collection channel.
Microfluidics sorting simplifies the final analysis of the droplets. Instead of analyzing a huge pool of droplets within which many of the droplets are empty, only a small population of the droplet will be analyzed all of which contain the target markers. -An original uFluidix Article.
Read more about what needs to be considered before designing a droplet microfluidic chip
Droplet microfluidics is one of the most popular microfluidic domains. Learn about its applications.