Designing a droplet microfluidic experiment: Tips on what to consider before you start
Setting up droplet microfluidic experiments can be tricky. There are a number of things that need to be considered in advance that the microfluidics chips are prepared for the experiments properly. In the following, we have put together a list of questions that need to be addressed before designing and fabricating a microfluidic droplet generator.
What droplet size is needed?
 There is no unique answer to this question. The required cell diameter is related to several parameters. The cell size is an important factor in determining the required droplet size. In some applications, there might be a need for incubating the cells within the droplets. Here, care should be taken to ensure the droplet volume is sufficient for containing the nutrients for the duration of the incubation. Each droplet generator microfluidic chip allows a range of droplet sizes to be produced stably. Outside of this range, either the droplets would not be uniform or no droplet would form. This span of droplet sizes is highly related to the microfluidic channel size and nozzle dimensions. The droplet size can be changed by changing the flow rates and flow rate ratios of the aqueous and oil phases.
There is no unique answer to this question. The required cell diameter is related to several parameters. The cell size is an important factor in determining the required droplet size. In some applications, there might be a need for incubating the cells within the droplets. Here, care should be taken to ensure the droplet volume is sufficient for containing the nutrients for the duration of the incubation. Each droplet generator microfluidic chip allows a range of droplet sizes to be produced stably. Outside of this range, either the droplets would not be uniform or no droplet would form. This span of droplet sizes is highly related to the microfluidic channel size and nozzle dimensions. The droplet size can be changed by changing the flow rates and flow rate ratios of the aqueous and oil phases.
What reagents and surfactants should be used?
The first thing that needs attention here is the reagents. The reagents can affect the droplet size and the vitality of the cells. At least one oil phase and one aqueous phase are needed for a droplet microfluidics experiment. The choice of the oil phase depends highly on the nature of the experiment. For example, in single-cell assays, it is very common to use fluorinated oil. The high capacity of fluorinated oils for oxygen increases cell vitality. Some common fluorinated oils are FC-40, FC-3283, and HFE-7500. Mostly, there is not as much flexibility as the oil phase in selecting the aqueous phase. For single-cell studies, it is dictated by the cell type and its nutritional demands whereas in material synthesis application it is the hydrogel type determines the composition of the aqueous phase.
Next, the surfactant needs to be determined. Surfactants are of utmost importance in droplet microfluidic experiments. Surfactants normally have a hydrophobic tail and a hydrophilic tail. They form a layer around the droplet which lowers the surface tension and increases the stability. Oftentimes, the droplets undergo several tasks after generation. A good surfactant ensures that the droplets do not merge during the passage through the microfluidic device. As the field grows and the experiments become more sophisticated, many researchers have turned to synthesize home-made surfactants that best suits their need. The proper choice of surfactant also prevents non-specific adsorption of the reagents. Some common surfactants for fluorinated oils include dimorpholinophosphate-PFPE (DM-PPFPE), polyethyleneglycol-PFPE (PEG-PFPE), and ammonium salt of carboxy-PFPE (Carboxy-PFPE).
What type of surface treatment is required for the microfluidics channels?
Polydimethylsiloxane (PDMS) is a popular material in microfluidic chips. PDMS is of a hydrophobic nature. However, upon exposure to plasma during plasma bonding, it temporarily becomes hydrophilic and reverts to hydrophobic in a few days. This feature is referred to as the hydrophobic recovery of the PDMS.
 Depending on the experiment, the requirements for hydrophobicity and hydrophilicity of the microchannel surface can be different. For oil in water droplets, a hydrophilic surface repels the droplets and prevents absorption or crawling of the droplets on the wall. This hydrophilic coating results in more stable and uniform oil in water droplets. For water in oil droplets, the case is the opposite. A hydrophobic surface is desirable for water in oil droplets. Although PDMS recovers to a hydrophobic state a few days after bonding, it is not hydrophobic enough. A hydrophobic coating is highly recommended in this case to reduce the contact angle of the surface and boosts the quality of the generated droplets.
Depending on the experiment, the requirements for hydrophobicity and hydrophilicity of the microchannel surface can be different. For oil in water droplets, a hydrophilic surface repels the droplets and prevents absorption or crawling of the droplets on the wall. This hydrophilic coating results in more stable and uniform oil in water droplets. For water in oil droplets, the case is the opposite. A hydrophobic surface is desirable for water in oil droplets. Although PDMS recovers to a hydrophobic state a few days after bonding, it is not hydrophobic enough. A hydrophobic coating is highly recommended in this case to reduce the contact angle of the surface and boosts the quality of the generated droplets.
Therefore, care should be taken to treat the surface of the channels according to the experiment. A proper surface treatment assures that the droplets will be stable and do not get disrupted by unnecessary interaction with the walls.
How do Poisson distribution and flow conditions affect my experiment?
The main questions over here are what flow rates and flow rate ratios should be used? And how much should be the concentration of the cells?
A good starting point for testing the flow rates can be deduced from other research articles with comparable channel dimensions. One can then change the flow rates and flow rate ratios and measure the droplet size at each point. A map can then be made that includes the droplet size vs. flow rate and flow rate vs. droplet generation frequency.
As the name implies, single-cell analysis studies, required the droplet not to contain more than one cell. However, the cells are randomly distributed inside the solution and thus the encapsulation process is random meaning that some droplets might contain more than one cell. The probability of having droplets with more than one cell significantly decreases when Poisson distribution formulas are considered. The Poisson distribution determines the concentration of the cells at which the probability of droplets with multiple cells falls below a particular threshold. Based on the threshold and the flow rates, the recommended cell concentration varies. Online Poisson calculators like the one developed by Dropletex can come in handy when designing a droplet microfluidic experiment for single-cell analysis.
What post-generation steps are required?
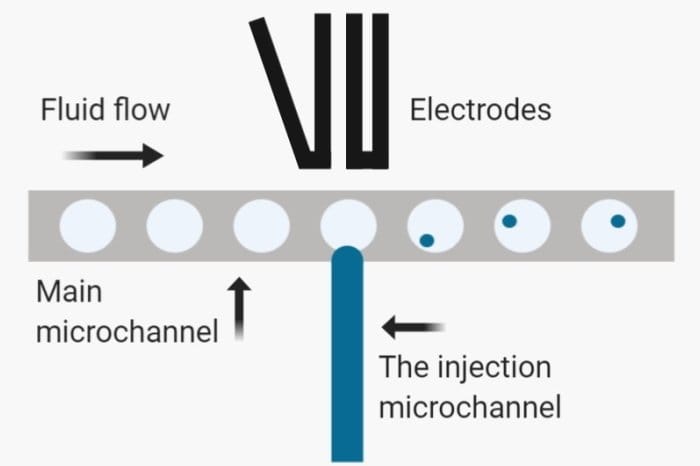
In many cases, the droplet formation and cell encapsulation do not suffice. Oftentimes, a post-generation step is needed to manipulate the droplets. This could include picoinjection, reinjection, incubation, thermocycling, polymerization, mixing, merging, splitting, etc. The microfluidics manipulator chip needs to be synced with the droplet generator chip in terms of the microfluidic channel dimension and surface treatment. -An original uFluidix Article.
Droplet microfluidics is one of the most popular microfluidic domains. Learn about its applications.
Read more about what needs to be considered before designing a droplet microfluidic chip