Passive microfluidic cell sorters as a tool for effective cell sorting at high resolution
Microfluidic cell sorters are microchips that employ the advantages of Microfluidics to sort and separate biological cells. Passive microfluidic cell sorters do not require any external power source for sorting the cells. The cell sorting in these Microfluidic chips is based on the forces exerted on cells in microchannels such as the inertial and Dean forces, or geometrical patterns.
Inertial microfluidics
Inertial microfluidic devices are probably one of the most popular passive microfluidic sorting platforms. It is getting very popular due to its simplicity, high-throughput, and precision. Inertial microfluidic cell sorters take advantage of two major forces that cells experience in microfluidic channels for separating the cells: Inertial force, and Dean force. This brings two classes of inertial microfluidic cell sorters, namely straight channel cell sorters and curved channel cell sorters.
Inertial microfluidic cell sorting in straight microchannels
Fluid flow in microchannels is associated with very low Reynolds numbers. Reynolds number is a dimensionless number that defines two things: The fluid regime in confined channels and the ratio of inertial forces to viscous (friction) forces. The lower the Reynolds number the less turbulent and chaotic the flow is. Also, a Reynolds number of larger than 1 causes the inertial forces to be considerable. 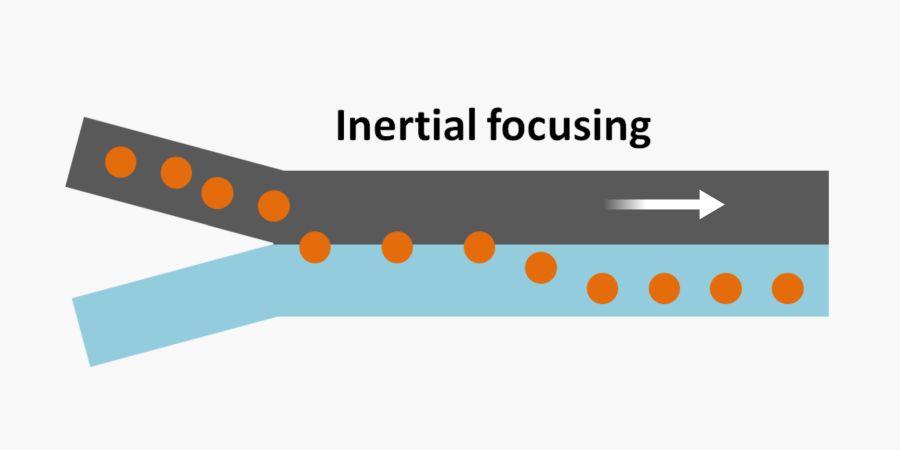 In inertial microfluidic chips, the Reynolds number is low enough for the fluid streams to be laminar and not chaotic or turbulent and high enough for inertial forces to be significant. As opposed to turbulent flows, laminar flow streams can be controlled and manipulated precisely. For example, the laminar flow allows two fluid streams to flow alongside each other without mixing.
In inertial microfluidic chips, the Reynolds number is low enough for the fluid streams to be laminar and not chaotic or turbulent and high enough for inertial forces to be significant. As opposed to turbulent flows, laminar flow streams can be controlled and manipulated precisely. For example, the laminar flow allows two fluid streams to flow alongside each other without mixing.
The cells and particles flowing inside straight microchannels experience an inertial force. The inertial force depends on cell size, shape, density, and fluid properties. Thus, the amount of inertial force that the cells experience while travelling inside the microchannels can vary from one cell to another depending on the cell properties. This force can cause the cells to migrate across the channel until they reach an equilibrium point. For instance, the cells that experience a larger force separate into a farther stream while the rest remain closer to the walls. This stable point depends on the cell properties and causes cells with different properties to differentiate across the microchannel. This differentiation is called inertial focusing. The chip needs to be long enough (Lmin) to ensure the cells have enough time to reach the equilibrium point. Outlet channels can then be strategically placed at the points of differentiation to collect the separated cells from different outlets.
Inertial microfluidic cell sorting in curved microfluidics channels
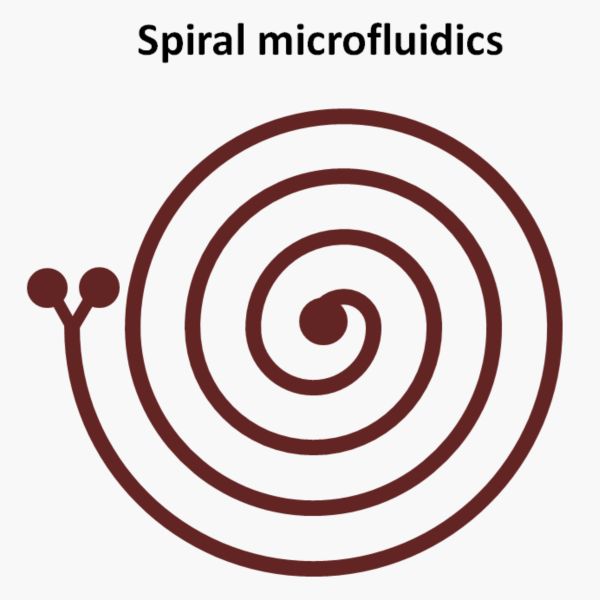 In curved microfluidics microchannels, other than the axial flow, another flow forms in the lateral direction called secondary flow or Dean flow. The centrifugal force in the curved channels results in a lateral pressure distribution which in turn leads to the formation of this Dean flow. The Dean flow usually appears in the form of two or multiple lateral counter-rotating vortices. The benefits of these Dean vortices in microfluidic sorting are many folds. They can help the cells and particles to reach the equilibrium faster that results in decreasing the length required for inertial focusing. The curved and spiral shape of the microfluidics chip also decreases the footprint of the device.
In curved microfluidics microchannels, other than the axial flow, another flow forms in the lateral direction called secondary flow or Dean flow. The centrifugal force in the curved channels results in a lateral pressure distribution which in turn leads to the formation of this Dean flow. The Dean flow usually appears in the form of two or multiple lateral counter-rotating vortices. The benefits of these Dean vortices in microfluidic sorting are many folds. They can help the cells and particles to reach the equilibrium faster that results in decreasing the length required for inertial focusing. The curved and spiral shape of the microfluidics chip also decreases the footprint of the device.
Here, other than the inertial force, another force called centrifugal force (Dean force) plays a role in determining the equilibrium point of the cells. Here, the ratio of the Dean force to the inertial force is the important parameter affecting the cell separation. Spiral microfluidics has caught attention in applications where high throughput and high flow rate is needed such as high throughput separation and enrichment of rare cell types for point of care purposes.
| Flow rate | Sheath-flow | Throughput | Design | |
| Straight Channel | Low-Medium | No | Low-Medium | Easy |
| Curved Channel | High | No | High | Moderate |
Deterministic Lateral Displacement:
 Deterministic Lateral Displacement (DLD) is well-known for its very fine resolution (down to 10nm) for cell separation. This class of passive microfluidics cell sorter relies on the geometrical features of the microchannel. These chips use an array of posts or pillars for cell separation. In Deterministic Lateral Displacement (DLD) chips, the cells that are larger than a critical diameter (Dc) displace laterally while the smaller cells do not change their path significantly. Each row shifts laterally with respect to the previous row. By this being repeated at each succeeding row, the distance between the large cells and smaller cells increases and they get separated. An array of pillars ensures the cells differentiate enough to be collected from different streams. Based on the pillar size, cells smaller than a critical size will move withing the initial streamline. The formulas for calculating the critical size, pillar size, and distance between pillars are well-documented and can be found in the papers in the reference section below. Multiple DLD arrays can be integrated to broaden the size separation span and separate a wider range of cell sizes. DLD microfluidics chips operate continuously and can be connected to larger systems as a modular unit for cell separation or purification.
Deterministic Lateral Displacement (DLD) is well-known for its very fine resolution (down to 10nm) for cell separation. This class of passive microfluidics cell sorter relies on the geometrical features of the microchannel. These chips use an array of posts or pillars for cell separation. In Deterministic Lateral Displacement (DLD) chips, the cells that are larger than a critical diameter (Dc) displace laterally while the smaller cells do not change their path significantly. Each row shifts laterally with respect to the previous row. By this being repeated at each succeeding row, the distance between the large cells and smaller cells increases and they get separated. An array of pillars ensures the cells differentiate enough to be collected from different streams. Based on the pillar size, cells smaller than a critical size will move withing the initial streamline. The formulas for calculating the critical size, pillar size, and distance between pillars are well-documented and can be found in the papers in the reference section below. Multiple DLD arrays can be integrated to broaden the size separation span and separate a wider range of cell sizes. DLD microfluidics chips operate continuously and can be connected to larger systems as a modular unit for cell separation or purification.
Microfluidics Microfiltration
 The simplest method for cell sorting is microfiltration. As the name suggests, here, the cells are sorted using filters embedded in the microfluidics channel. The filters allow cells smaller than a particular size to pass and filter larger cells. Many filtration microfluidic devices are fabricated to include weirs, pillars, and cross-flow. In the weir approach, cells encounter a weir in the channel. The weir traps the large cells behind and allows smaller cells to pass through the top gap. Pillars can also filter large cells and let small cells to escape from the gap between them. Cross-flow is similar to pillars but the filter is placed laterally. Here, the pillars at the entrance of a side-channel allow small cells to pass while the main flow carries the large cells to another outlet. Similar to DLD, cross-flow, and pillar filtration both use pillars. However, they should not be confused since in DLD the pillar sizes and gaps are designed such that cells follow a particular streamline. But, in pillar and cross-flow filtration the pillars trap the large cells and do not let them pass.-An original uFluidix content.
The simplest method for cell sorting is microfiltration. As the name suggests, here, the cells are sorted using filters embedded in the microfluidics channel. The filters allow cells smaller than a particular size to pass and filter larger cells. Many filtration microfluidic devices are fabricated to include weirs, pillars, and cross-flow. In the weir approach, cells encounter a weir in the channel. The weir traps the large cells behind and allows smaller cells to pass through the top gap. Pillars can also filter large cells and let small cells to escape from the gap between them. Cross-flow is similar to pillars but the filter is placed laterally. Here, the pillars at the entrance of a side-channel allow small cells to pass while the main flow carries the large cells to another outlet. Similar to DLD, cross-flow, and pillar filtration both use pillars. However, they should not be confused since in DLD the pillar sizes and gaps are designed such that cells follow a particular streamline. But, in pillar and cross-flow filtration the pillars trap the large cells and do not let them pass.-An original uFluidix content.
Further Reading
- Inertial microfluidics
- Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics
- Microfluidic curved-channel centrifuge for solution exchange of target microparticles and their simultaneous separation from bacteria
- Label-free mesenchymal stem cell enrichment from bone marrow samples by inertial microfluidics
- Liquid biopsy for minimal residual disease detection in leukemia using a portable blast cell biochip
- A microfluidic chip integrated with a high-density PDMS-based microfiltration membrane for rapid isolation and detection of circulating tumor cells
- Tangential Flow Microfiltration for Viral Separation and Concentration
- A multilayer lateral-flow microfluidic device for particle separation
- Sequestration of bacteria from whole blood by optimized microfluidic cross-flow filtration for Rapid Antimicrobial Susceptibility Testing
Read more on how active microfluidic cell sorters are used for efficient and highly accurate sorting of biological cells using microfluidics principles