Microfluidic Electrokinetics for Cell Characterization: A Review
What Does Electrokinetics Mean?
Electrokinetics describes the motion of particles and/or fluids under applied E-fields. Electrokinetics has been widely used in Microfluidics to characterize biological objects such as cells. Here we a look at different categories of electrokinetics used in microlfuidic devices. These include Electrophoresis, Electroosmosis, Dielectrophoresis, Traveling Wave Dielectrophoresis, and Electrorotation. Key research work in each of these categories and their applications to cell-based Microfluidics innovations is also presented.
What is Electrophoresis on a Chip?
Electrophoresis is the motion of a charged particle due to action of Coulomb force. Electrophoretic force depends on surface charge of the particle and the strength of the acting electric field (E-field)(Fig(a)). Cells suspended in an electrolyte typically have negative charge on the surface under normal physiological conditions at pH 7, and so they can be manipulated in the E-field. Practical applications of electrophoresis mostly involve cells suspended in an uniform E-field, where the dimension of the cells are small compared to the length scale of the variation distance in the field strength. Measurement of the electrophoretic responses of cells freely suspended in solution represented one of the earliest of analytical methods to study the surface properties of cells and especially membrane surface charge[1]. In methods known as free-flow-electrophoresis or microelectrophoresis, an E-field is established in a rectangular or cylindrical cuvette or chamber of a microfluidic chip. Then with the aid of a microscope camera the time taken for a cell to move a defined distance can be determined. Similarly, the deviation of a cell from a vertical fluid flow path (caused by applying an E-field perpendicular to the flow stream) may also be measured [2]. Electrophoresis has also been developed as a preparative method to separate cell subpopulations from cell mixtures[3]. Electrophoresis for separation of cells is less efficient than some other microfluidics methods such dielectrophoresis.
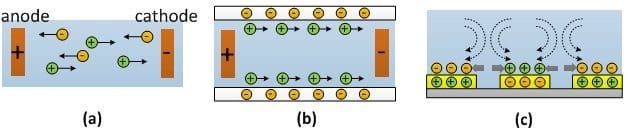
(a) Electrophoresis, (b) DC electroosmosis, (c) AC electroosmisis. The dotted arrows represent the fluid motion while the solid lines indicate the motion of the particles suspended in the fluid.
What is Electroosmosis?
Direct current (DC) Electroosmotic (EO) flow is the motion of a liquid that contains a net charge, typically as a result of proximity to a solid surface and formation of an electric double layer (EDL) at the interface. In this method a one of the layers is bound to the surface, while the layer with opposite polarity remains in the liquid. The layer away from the wall is more mobile and thus would move under appropriate electrical field (Fig. (b), above). In microchannels, EO creates a plug flow where the velocity outside the double layer remains constant, which is different from the parabolic flow profile caused by pressure or volume driven flow.This phenomenon can be used for pumping the cell suspension into and through microfluidic devices, but generally not for finer manipulation or modification of the suspended cells.
Recently, alternative current (AC) EO was demonstrated where the applied E-field acts on its own induced charges at the electrode surfaces to create fluid flow. This type of microfluidics devices generally require much smaller operational voltages (in mV) and therefore undesirable electrochemical reactions are reduced or eliminated. The two sets of electrodes in AC approach create a pair of counter rotating vortices which focuses and traps cells and particles on top of the electrodes (Fig. (c)). AC EO using micropatterned electrode arrays has been applied to particle focusing and concentration in Microfluidic chips [4, 5].
What is Dielectrophoresis (DEP) on Chip?
Dielectrophoresis (DEP) refers to the interaction force between a non-uniform E-field and the dipole moment it induces on a 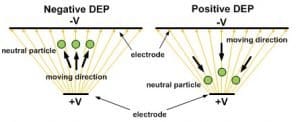 polarizable object. The magnitude of DEP force, (FDEP) on a spherical particle strongly depends on permittivity of the medium surrounding the sphere, volume of the bio-particle, gradient of E-field strength, and real-part of Clausius-Mossotti factor (CM*). CM* is a complex variable related to the complex permittivity of the particle and the medium. For a particle more polarizable than the medium, the real part of its CM* factor is positive: Re[CM* (ω)]>0 . This Means the target experiences a positive DEP (pDEP) force moving it toward the strong E-field region. On the other hand, if Re[CM* (ω)]<0, the particle experiences a negative DEP (nDEP) force moving it to the weak E-field region. The detailed and analytical models of Clausius-Mossotti factor and approximated DEP force can be found in [6].
polarizable object. The magnitude of DEP force, (FDEP) on a spherical particle strongly depends on permittivity of the medium surrounding the sphere, volume of the bio-particle, gradient of E-field strength, and real-part of Clausius-Mossotti factor (CM*). CM* is a complex variable related to the complex permittivity of the particle and the medium. For a particle more polarizable than the medium, the real part of its CM* factor is positive: Re[CM* (ω)]>0 . This Means the target experiences a positive DEP (pDEP) force moving it toward the strong E-field region. On the other hand, if Re[CM* (ω)]<0, the particle experiences a negative DEP (nDEP) force moving it to the weak E-field region. The detailed and analytical models of Clausius-Mossotti factor and approximated DEP force can be found in [6].
DEP can be used to manipulate, characterize and separate cells in a label free and noninvasive method. In microfluidics or Lab-on-a-Chip applications, DEP has prominent effects to create strong E-field gradients due to the micro-scale distances from electrodes or insulating structures distorting the E-field in microfluidic channels. The typical DEP forces are in the range of 10’s of pico-Newton. However, the major drawback of using DEP for practical application is to re-suspend cells in a low conductivity buffer (10’s to 100’s µS/m) to mitigate Joule heating rather than direct operation in high conductivity regular physiological buffer (1~2 S/m).Typically, DEP can be used for high-throughput or high-speed cell manipulation in continuous microfluidic flows[7, 8], performing single-cell characterization of dielectric signatures (Re[CM*])[9]. IT is also used for cell separation based on cell’s biophysical properties[10]. DEP has not only been a popular research topic, in the last few years DEP-based devices have become commercially available as well[11].
What is Microfluidic Electrorotation?
Electrorotation (EROT) is commonly used as a tool to characterize electrical properties of single cells, such as Im[CM*], cell conductivity, and permittivity. Compared to DEP, EROT focuses more on the cell characterization of Im[CM*] compared to Re[CM*] in DEP. A typical quadrupole electrode layout used in microfluidic devices for EROT is illustrated in Fig(a). In this layout electrodes are placed around a cell. 90ophase changes are then applied to the electrodes to generate a rotational field. As such, an induced dipole in the middle of the electrodes will experience torque and rotate particles such as cells. Detailed derivation of the TROT forces could be found in [6].The cells can rotate in the same direction or opposite direction with the E-field, depending on the E-field excitation frequency f and Im[CM*]. The EROT microfluidic devices are usually designed to have an area of constant field gradient in the middle between the electrodes Fig.(a). This is to make it easier to determine or calculate the field around the cell. A more advanced microfluidics setup uses combination of EROT and nDEP. In this method EROT and nDEP excitation signals can be super-positioned to trap the cell in the middle of the quadrupole electrodes as well as performing EROT characterization[12, 13]. For a different application, the rotational velocity of individual cells can be measured by recording videos followed by image analysis. From the velocity measured at different frequencies, the imaginary part of the Clausius–Mossotti factor can be fitted to the measurements to calculate electrical conductivities and permittivity of cells. However, the major drawback of EROT is its extremely low throughput, usually at couple cells per hour.
What is Traveling Wave Dielectrophoresis
Traveling wave dielectrophoresis (twDEP) is simply a linear application of EROT in Microfluidics. An AC electric wave is produced by applying an E-field that travels linearly along a series of electrodes. A typical example of a twDEP system with a 90◦ phase shift between interdigitated electrodes is illustrated in Fig.(b). Detailed derivation of the twDEP forces could be found in [14]. 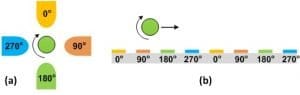 T he twDEP forces incorporate the imaginary part of the CM*; the direction of the particle with respect to the traveling wave is dependent on this factor. A cell can effectively move if the DEP force is nDEP in order to levitate the particle above the electrode surface and at the same time the twDEP component is large enough to produce translational motion. In order to implement twDEP, metal electrodes need to be deposited in multiple layers with proper insulation while fabricating the microfluidic device. Alternatively a manifold can be designed to individually connect each electrode stripes of the interdigitated electrode array. Compared to DEP, the major advantage of twDEP is to transport cells on-chip without the need of bulky pressure sources or syringe pumps to create liquid flows. This particularly fits the needs of miniaturized Lab-on-a-Chip applications and small volume sample processing. In addition, twDEP can achieve more than just induced controlled translational motion, such as separating particles of different dielectric properties [15] or integrating DEP + twDEP + EROT for single-cell diagnosis [16].
T he twDEP forces incorporate the imaginary part of the CM*; the direction of the particle with respect to the traveling wave is dependent on this factor. A cell can effectively move if the DEP force is nDEP in order to levitate the particle above the electrode surface and at the same time the twDEP component is large enough to produce translational motion. In order to implement twDEP, metal electrodes need to be deposited in multiple layers with proper insulation while fabricating the microfluidic device. Alternatively a manifold can be designed to individually connect each electrode stripes of the interdigitated electrode array. Compared to DEP, the major advantage of twDEP is to transport cells on-chip without the need of bulky pressure sources or syringe pumps to create liquid flows. This particularly fits the needs of miniaturized Lab-on-a-Chip applications and small volume sample processing. In addition, twDEP can achieve more than just induced controlled translational motion, such as separating particles of different dielectric properties [15] or integrating DEP + twDEP + EROT for single-cell diagnosis [16].
References and Further Reading
- Mehrishi, J.N. and J. Bauer, Electrophoresis of cells and the biological relevance of surface charge. Electrophoresis, 2002. 23(13): p. 1984-1994.
- Breadmore, M.C., et al., Recent advances in enhancing the sensitivity of electrophoresis and electrochromatography in capillaries and microchips (2016 – 2018). Electrophoresis, 2018. 40(1): p. 17-39.
- Roman, M.C. and P.R. Brown, Free-flow electrophoresis as a preparative separation technique. Analytical Chemistry, 1994. 66(2): p. 86A-94A.
- PrashantTathireddy, Yunn-HongChoi, and MikhailSkliar, Particle AC electrokinetics in planar interdigitated microelectrode geometry. Journal of Electrostatics, 2008. 66(11-12): p. 609-619.
- Bown, M.R. and C.D. Meinhart, AC electroosmotic flow in a DNA concentrator. Microfluid Nanofluid, 2006. 2: p. 513-523.
- Jones, T.B., Electromechanics of Particles. 1995: Cambridge University Press.
- Kung, Y.C., et al., Tunnel Dielectrophoresis for Tunable, Single‐Stream Cell Focusing in Physiological Buffers in High‐Speed Microfluidic Flows. Small, 2016. 12(32): p. 4343-4348.
- Kung, Y.-C., et al., Fabrication of 3D high aspect ratio PDMS microfluidic networks with a hybrid stamp. Lab on a Chip, 2015. 15(8): p. 1861-1868.
- Jaffe, A. and J. Voldman, Multi-frequency dielectrophoretic characterization of single cells. Microsystems & Nanoengineering, 2018. 4(23).
- Wei, Z., et al., Flow-Through Cell Electroporation Microchip Integrating Dielectrophoretic Viable Cell Sorting. Analytical Chemistry, 2014. 86(20): p. 10215-10222.
- Trapani, M.D., N. Manaresi, and G. Medoro, DEPArray™ system: An automatic image‐based sorter for isolation of pure circulating tumor cells. Cytometry Part A, 2018. 93(12): p. 1260-1266.
- Han, S.-I., Y.-D. Joo, and K.-H. Han, An electrorotation technique for measuring the dielectric properties of cells with simultaneous use of negative quadrupolar dielectrophoresis and electrorotation. Lab on a Chip, 2013. 138(5): p. 1529-1537.
- Kelbauskas, L., et al., Optical computed tomography for spatially isotropic four-dimensional imaging of live single cells. Science Advances, 2017. 3(12): p. e1602580.
- Hughes, M.P., R. Pethig, and X.-B. Wang, Dielectrophoretic forces on particles in travelling E-fields. Journal of Physics D: Applied Physics, 1995. 29(2): p. 474-482.
- Cheng, I.-F., et al., A continuous high-throughput bioparticle sorter based on 3D traveling-wave dielectrophoresis. Lab on a Chip, 2009. 9(22): p. 3193-3201.
- Cen, E.G., et al., A combined dielectrophoresis, traveling wave dielectrophoresis and electrorotation microchip for the manipulation and characterization of human malignant cells. Journal of Microbiological Methods, 2004. 58(3): p. 387-401.
Use electric, magnetic or accoustic forces to sort and separate cells on microfluidic devices.