28 Feb Microfluidic Platform for Automated Organoid Culture and Longitudinal Imaging
Organoids have transformed in vitro tissue modeling, but their culture remains labor-intensive and variable. Manual media exchange, orbital shakers, and dependence on humidified CO₂ incubators make continuous monitoring difficult and introduce experimental inconsistencies. Although microfluidic systems improve media exchange and reproducibility, most still rely on conventional incubators, limiting long-term imaging and integration of electronics. In this work, the authors present a modular microfluidic device that integrates automated feeding, environmental control, and longitudinal imaging into a single, incubator-independent system
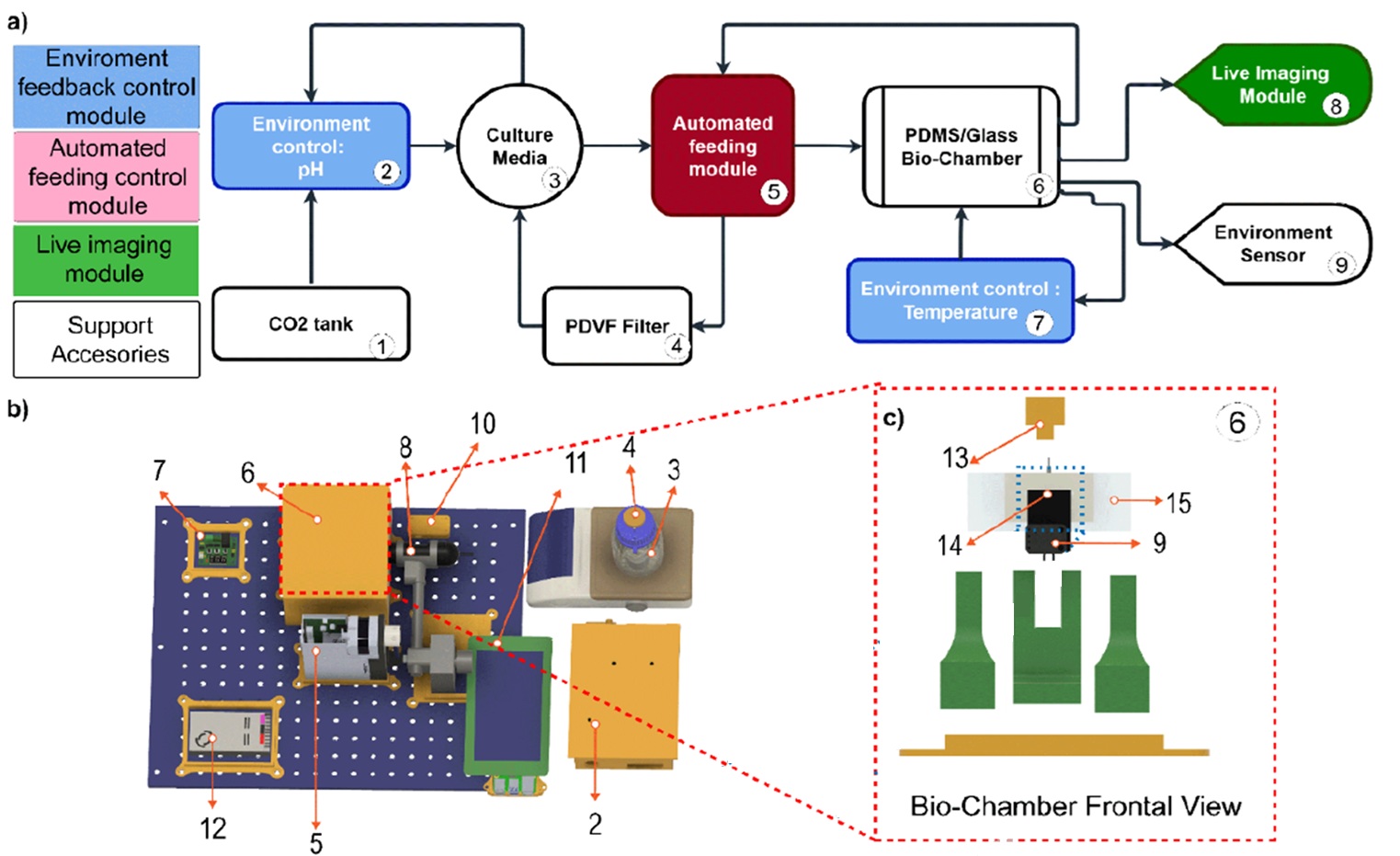
To address the limitations of traditional organoid culture, the authors developed a three-module system composed of automated media feeding, live imaging acquisition, and environmental feedback control. A defining feature of the platform is a vertically oriented PDMS/glass microfluidic chip that enables controlled perfusion while allowing direct visualization of fluid flow approaching the organoid. The system regulates temperature and pH in real time, eliminating the need for a standard incubator and enabling stable long-term monitoring

“Overview of the modular platform for automated organoid culture and longitudinal imaging. a) Block diagram showing the main modules: environmental control (2, 7), automated feeding (5), and live imaging (8). b) CAD model of the whole system: heater controller (7), biochamber (6, highlighted in red and detailed in panel c), portable microscope (8), environmental sensor control module (10), LED visualization station (11), media reservoir with pH probe (3), pH controller (2), programmable servo pump (5), and power supply (12). c)” Reproduced from Torres-Montoya, S., Hernandez, S., Seiler, S.T. et al. A modular platform for automated organoid culture and longitudinal imaging. Sci Rep (2026). under a Creative Commons Attribution 4.0 International License.
The microfluidic platform was built using off-the-shelf 3D-printed components mounted on an optical breadboard. The environmental control module includes a pH feedback loop that injects CO₂ into the media and a temperature control unit capable of maintaining 37°C within ±0.6°C and pH stability within ±0.3 units. The automated feeding module uses a programmable servo pump with 5 nL resolution and adjustable stroke durations from 1 second to 5 hours, supporting semi-continuous recirculation to enhance nutrient exchange. Media was dispensed at 53 μL and aspirated at 50 μL every 60 seconds to compensate for evaporation.
The PDMS/glass microfluidic chip was microfabricated by bonding molded PDMS wells to microscope slides using oxygen plasma activation. Inlet and outlet channels were positioned 5 mm above the well bottom to prevent organoid loss. The chip was oriented vertically to enable real-time tracking of perfusion profiles. Fabrication required less than 16 hours, and the material cost per chip was approximately $5.31. Imaging was performed using a fluorescence-capable digital microscope, allowing brightfield and fluorescent acquisition, including GFP and mCherry detection.
To validate biological compatibility, mouse cerebral organoids were cultured on the platform and compared to incubator-grown controls. After six days, organoids maintained comparable viability and cytoarchitecture, confirmed by CytoPainter membrane staining and Map2 immunohistochemistry. Metabolic profiling using the Vi-CELL MetaFLEX analyzer showed no statistically significant differences in pH, glucose, sodium, potassium, calcium, or chloride concentrations between platform and incubator conditions, confirming metabolic stability across three independent batches.
Longitudinal brightfield imaging demonstrated consistent organoid growth from day 15 to day 20. Cross-sectional area measurements revealed reproducible expansion across batches, with statistically significant growth trends confirmed by Kruskal-Wallis testing. Multiwell chip configurations reduced organoid fusion and improved spatial separation compared to traditional culture formats.
To further demonstrate microfluidic control, the authors performed a real-time dye absorption experiment using BODIPY under pulsatile flow. Fluorescence intensity was tracked in defined regions of interest within the organoid. The measured concentration profiles closely matched computational fluid dynamics simulations, confirming accurate synchronization between experimental perfusion dynamics and numerical modeling. This capability highlights the platform’s potential for studying dynamic morphogen gradients, drug delivery, and tissue permeability in real time.
In conclusion, this modular microfluidic platform integrates automated perfusion, environmental regulation, and live imaging into a unified organoid culture system. By removing the constraints of conventional incubators and enabling longitudinal tracking of growth, metabolism, and media transport, the system provides a versatile framework for disease modeling, drug screening, and developmental studies. The combination of controlled microfluidic circulation with computational validation positions this platform as a scalable tool for advanced organoid research.
Figures are reproduced from Torres-Montoya, S., Hernandez, S., Seiler, S.T. et al. A modular platform for automated organoid culture and longitudinal imaging. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40231-0 under a Creative Commons Attribution 4.0 International License.
Read the original article: A modular platform for automated organoid culture and longitudinal imaging
For more insights into the world of microfluidics and its burgeoning applications in biomedical research, stay tuned to our blog and explore the limitless possibilities that this technology unfolds. If you need high quality microfluidics chip for your experiments, do not hesitate to contact us.


