09 Apr Automated Microinjection of Zebrafish Embryos Using Magnetic Tweezers
Microinjection is a key technique for delivering genetic material into cells, especially in model organisms like zebrafish. However, traditional manual microinjection is slow, highly dependent on operator skill, and prone to variability. It also struggles with controlling cell orientation, which is critical to avoid damaging important cellular structures during injection. As demand grows for high-throughput and reproducible experiments in developmental biology, manual approaches become a major bottleneck
The authors developed an automated batch microinjection system that integrates a microfluidic chip with magnetic tweezers and computer vision. The microfluidic device enables precise control over cell positioning, orientation, and injection in a continuous workflow. By combining microfluidic transport with magnetic manipulation, the platform can reorient cells in three dimensions before injection, improving both accuracy and consistency

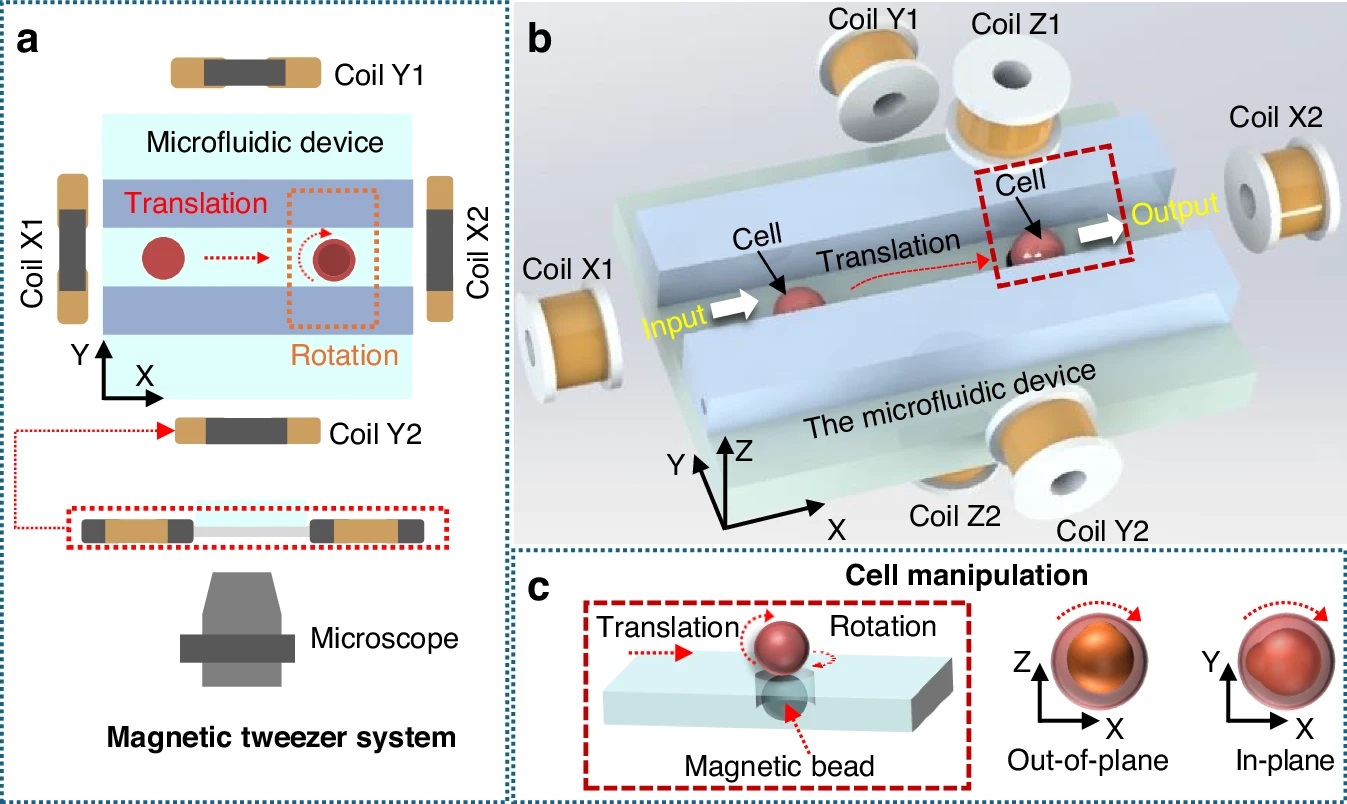
“a Two-dimensional magnetic tweezers system based on a microfluidic device under a microscope. b Three-dimensional magnetic tweezers system, including four magnetic coils in the X-Y plane and two vertical coils in the Z direction. Cell transportation and reorientation. c Schematic diagram of cell rotation outside the plane and rotation within the plane. In (b, c), the red box region corresponds to the area where the magnetic beads are located directly beneath the cell” Reproduced from Guo, X., Wang, F., Zhao, A. et al. Design of an automated cell batch microinjection system based on magnetic tweezers for zebrafish embryos. Microsyst Nanoeng 12, 113 (2026). https://doi.org/10.1038/s41378-026-01230-3 under a Creative Commons Attribution 4.0 International License.
The system is built around a multifunctional microfluidic chip that includes distinct regions for cell transport, reorientation, and injection. Cells are moved through microchannels using liquid flow, then held in place using gas pressure during injection. Magnetic beads positioned near the cells are actuated by a 3-dimensional Helmholtz coil setup, allowing controlled rotation of the cells via magnetic torque.
This magnetic actuation enables both in-plane and out-of-plane rotation, ensuring that each embryo is aligned correctly before puncture. The injection itself is performed using a microforce-controlled injector integrated into the microfluidic system, minimizing mechanical damage.
To guide the process, the authors designed a deep learning-based perception system called the microscopic manipulation perception network. This model detects cells and microneedles while also segmenting cellular regions, enabling precise targeting within a limited microscope field of view. The system processes image feedback in real time to coordinate positioning, orientation, and injection steps
The microfluidic system demonstrated strong performance across several metrics. Detection accuracy reached 98.8%, while segmentation accuracy was 98.4%, enabling reliable visual guidance during operation. The average operation time per cell was reduced to 33.8 seconds, compared to over 80 seconds for manual injection, representing a 2.3× improvement in efficiency.
Cell orientation error was limited to 2.1°, and the puncture success rate reached 100%. Importantly, the system maintained a cell survival rate of 88%, which is higher than typical manual approaches. Fluorescent imaging of zebrafish larvae confirmed successful gene expression, validating the effectiveness of the injection process
This work presents a fully integrated microfluidic platform for automated cell microinjection that combines magnetic manipulation, fluid control, and machine learning. By enabling precise cell orientation and reducing manual intervention, the system improves both throughput and reproducibility. The results show strong potential for applications in genetic studies, drug screening, and developmental biology, and the approach can be extended to other cell types with appropriate design adjustments.
Figures are reproduced from Guo, X., Wang, F., Zhao, A. et al. Design of an automated cell batch microinjection system based on magnetic tweezers for zebrafish embryos. Microsyst Nanoeng 12, 113 (2026). https://doi.org/10.1038/s41378-026-01230-3 under a Creative Commons Attribution 4.0 International License.
Read the original article: Design of an automated cell batch microinjection system based on magnetic tweezers for zebrafish embryos
For more insights into the world of microfluidics and its burgeoning applications in biomedical research, stay tuned to our blog and explore the limitless possibilities that this technology unfolds. If you need high quality microfluidics chip for your experiments, do not hesitate to contact us.


