10 Sep Microfluidics reveals the role of olfactory neurons in mechanosensitivity in C. elegans
Since its emergence as a model organism, Caenorhabditis elegans (C. elegans), has appeared as a powerful means for studying a broad range of topics ranging from developmental biology to neuroscience. Having many genes with functional counterparts in humans, a transparent body, a short life cycle of two weeks, small size, and being easy to mutate are among the advantages of using C. elegans as a model organism. While remaining a primitive and simple model organism, C. elegans carries some complex circuits such as the insulin signalling pathway. Microfluidics enables precise handling of fluids at micron-scale and thus has been extensively used in the past several years for studying C. elegans under controlled conditions in the so-called worm-on-a-chip (also called worm chips). In this week’s research highlight, we will introduce a recent research article published in Advanced Biology journal where a research team from Italy employs a microfluidic chip to investigate the mechanosensitivity of Amphid wing “C” (AWC) olfactory neurons in C. elegans.
Abstract
“AWC olfactory neurons are fundamental for chemotaxis toward volatile attractants in Caenorhabditis elegans. Here, it is shown that AWCON responds not only to chemicals but also to mechanical stimuli caused by fluid flow changes in a microfluidic device. The dynamics of calcium events are correlated with the stimulus amplitude. It is further shown that the mechanosensitivity of AWCON neurons has an intrinsic nature rather than a synaptic origin, and the calcium transient response is mediated by TAX-4 cGMP-gated cation channel, suggesting the involvement of one or more “odorant” receptors in AWCON mechanotransduction. In many cases, the responses show plateau properties resembling bistable calcium dynamics where neurons can switch from one stable state to the other. To investigate the unprecedentedly observed mechanosensitivity of AWCON neurons, a novel microfluidic device is designed to minimize the fluid shear flow in the arena hosting the nematodes. Animals in this device show reduced neuronal activation of AWCON neurons. The results observed indicate that the tangential component of the mechanical stress is the main contributor to the mechanosensitivity of AWCON. Furthermore, the microfluidic platform, integrating shearless perfusion and calcium imaging, provides a novel and more controlled solution for in vivo analysis both in micro-organisms and cultured cells.”
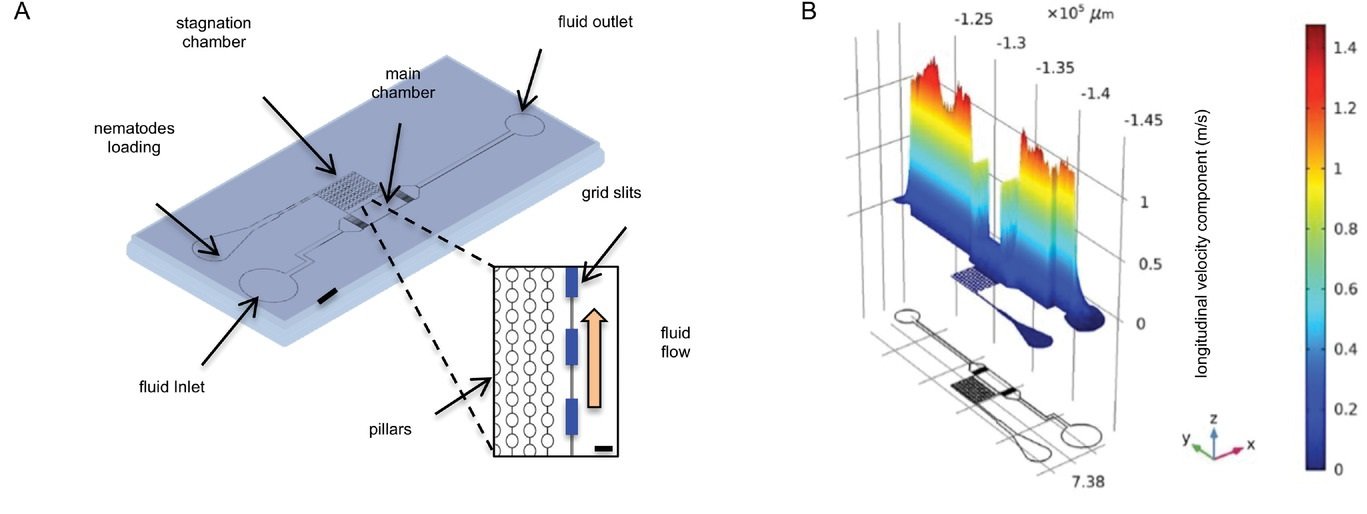
The working mechanism of the microfluidic device
Here, the research group hypothesized that exposing C. elegans to olfactory stimulants under flow conditions can also induce unwanted mechanosensitive processes. Therefore, a microfluidic device was designed to distinguish between the two by dividing the microfluidic channels into two compartments. The proposed microfluidic chip is made with PDMS with a thickness of 50 µm. Conventional soft lithography and photolithography techniques were used for the microfabrication of microfluidic chips. The nematodes are loaded from the nematodes loading inlet and get trapped in the stagnation chamber. The fluid flow is introduced to the main chamber via the fluid inlet where the grid slits, optimized through simulations, prevent the propagation of the mechanical stress variation to C. elegans. This design enabled the research team to isolate the effect of the hydrostatic pressure from the mechanical force due to the flow. The nematodes were then exposed to repeated mechanical stimulation and the neuronal response was recorded via calcium imaging.
Reproduced under Creative Commons Attribution 4.0 International License. Kwak et al., Sci. Rep., 2021.
“Our results contribute to shed light on the molecular mechanisms underpinning the complex nature of the C. elegans polymodal sensory neurons. Moreover, the microfluidic platform we employ can be adapted for the study of cell cultures as well as for on-chip experiments in which preventing the influence of mechanical stimuli is a critical aspect. In fact, when working with microfluidic devices, the shear stress experienced by the cells is a fundamental factor to take into consideration.”, the authors concluded.
Read the original article: A Shearless Microfluidic Device Detects a Role in Mechanosensitivity for AWCON Neuron in Caenorhabditis elegans

Pouriya Bayat
Pouriya is a microfluidic production engineer at uFluidix. He received his B.Sc. and M.A.Sc. both in Mechanical Engineering from Isfahan University of Technology and York University, respectively. During his master's studies, he had the chance to learn the foundations of microfluidic technology at ACUTE Lab where he focused on designing microfluidic platforms for cell washing and isolation. Upon graduation, he joined uFluidix to even further enjoy designing, manufacturing, and experimenting with microfluidic chips. In his free time, you might find him reading a psychology/philosophy/fantasy book while refilling his coffee every half an hour. Is there a must-read book in your mind, do not hesitate to hit him up with your to-read list.


