24 Jan Study of Eccrine Sweat Gland Development Using Microfluidic Chips Fabricated at uFluidix
Eccrine sweat glands are vital for human thermoregulation and, like other mammalian skin appendages, originate from multipotent epidermal progenitors. This research, leveraging advanced microfluidic technology, offers fascinating insights into the intricacies of eccrine gland formation and its potential applications in dermatology and regenerative medicine.
“Our study defines the signatures of eccrine identity and uncovers the eccrine dermal niche, setting the stage for targeted regeneration and comprehensive skin repair“, the authors explained.
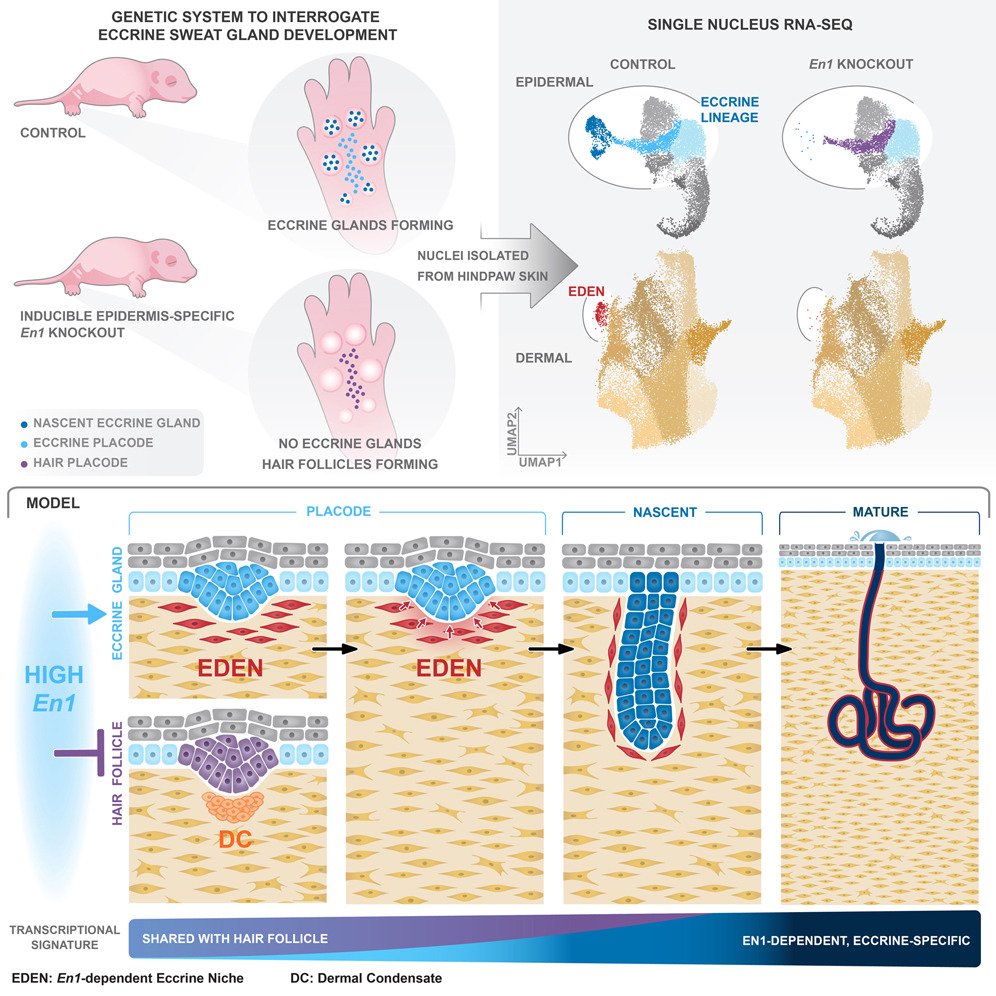
The study’s cornerstone lies in its use of a microfluidic chip which was microfabricated here at uFluidix microfabrication facilities. The microfluidic device enabled the researchers to conduct detailed analyses of epidermal progenitors. They discovered two concurrent epidermal transcriptomes in the early stages of eccrine development. One is shared with hair follicles, while the other is En1-dependent and specific to eccrine glands. This dual pathway is crucial for understanding the specialization process of epidermal progenitors towards eccrine gland formation.
The research applied single-nucleus transcriptomics to compare expression in normal eccrine-forming mouse skin with mice having a skin-specific disruption of the Engrailed 1 (En1) gene, which is a key factor in eccrine gland formation. The study revealed two concurrent epidermal transcriptomes in early eccrine development: one shared with hair follicles and an En1-dependent transcriptome specific to eccrine glands. It was shown that eccrine development requires the induction of a dermal niche proximal to each developing gland, a finding that is crucial for both understanding eccrine identity and for potential targeted regeneration and comprehensive skin repair.

The study found that En1-dependent dermal populations, associated with developing eccrine glands, come from a single lineage. This lineage, identified through single-nucleus RNA sequencing (snRNA-seq) and subclustering, was necessary for eccrine gland formation. The researchers utilized cell-cell interaction modeling to demonstrate significant receptor-ligand interactions between the eccrine-associated dermal and epidermal subclusters, indicating a bidirectional crosstalk essential for eccrine gland development. This interaction pattern was confirmed through an experiment where genetically ablating S100a4-expressing cells in the dermis led to a significant reduction in eccrine gland density, highlighting the necessity of the En1-dependent dermal population for eccrine gland formation.
Reflecting on Charles Darwin’s observations, the study underlines the physiological importance of eccrine glands and presents a blueprint for their development. It identifies key transcriptional and cellular transitions where the developmental paradigm of ectodermal appendages diverges to form eccrine glands.
The advancements in microfluidics, as showcased in this study, are revolutionizing our approach to understanding complex biological processes. The precision and control offered by microfluidic devices, a product of meticulous microfabrication, are indispensable tools in unraveling the secrets of human physiology and disease.
For more insights into the world of microfluidics and its burgeoning applications in biomedical research, stay tuned to our blog and explore the limitless possibilities that this technology unfolds. If you need high quality microfluidics chip for your experiments, do not hesitate to contact us.
Figures are reproduced from Heather L. Dingwall, Reiko R. Tomizawa, Adam Aharoni, Peng Hu, Qi Qiu, Blerina Kokalari, Serenity M. Martinez, Joan C. Donahue, Daniel Aldea, Meryl Mendoza, Ian A. Glass, Hao Wu, Yana G. Kamberov, Sweat gland development requires an eccrine dermal niche and couples two epidermal programs, Developmental Cell, Volume 59, Issue 1, 2024, Pages 20-32.e6,. https://doi.org/10.1016/j.devcel.2023.11.015 under a CC BY-NC-ND 4.0 DEED license.
Read the original article: Sweat gland development requires an eccrine dermal niche and couples two epidermal programshttps://creativecommons.org/licenses/by-nc-nd/4.0/


