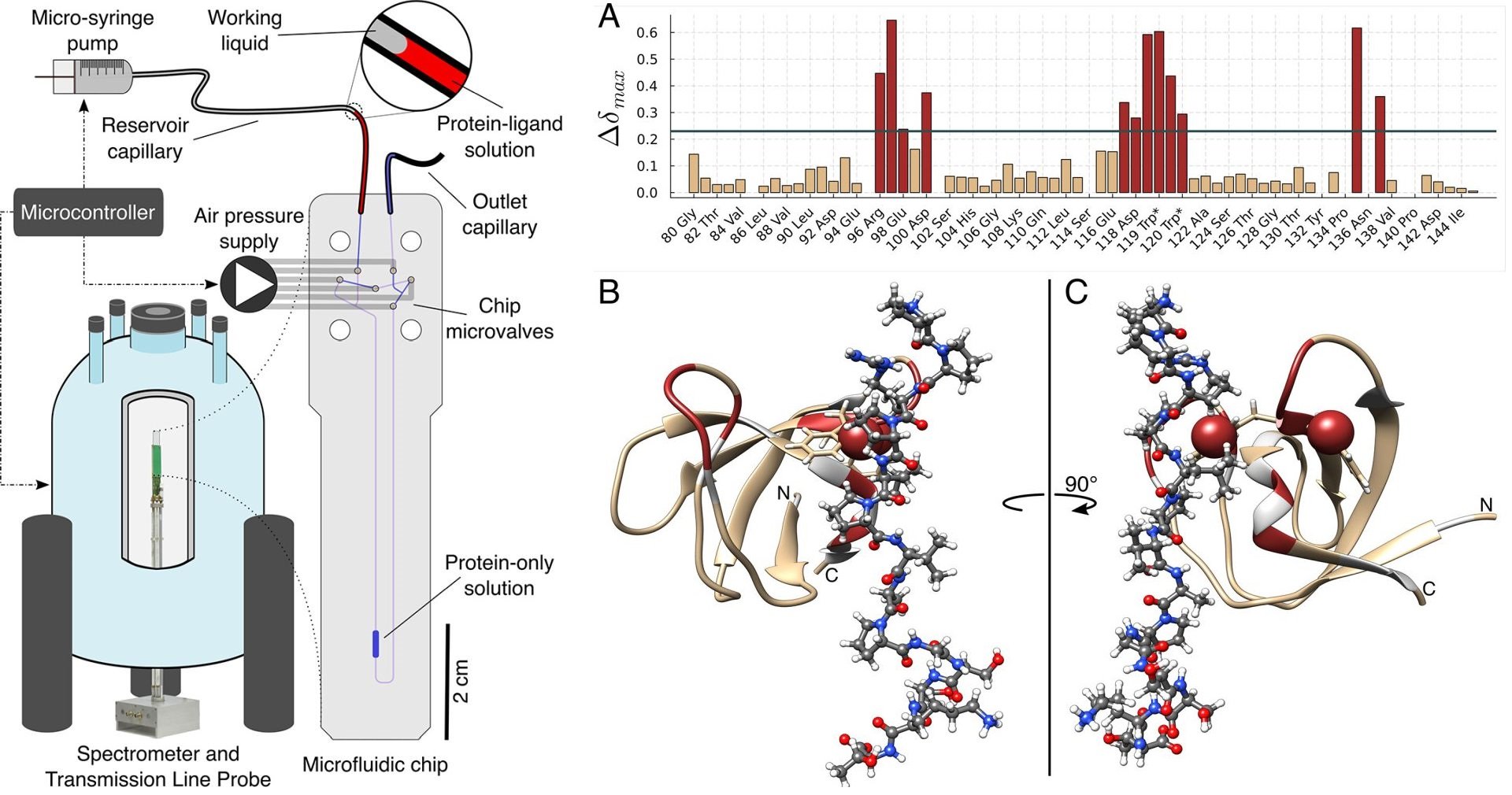
24 Feb Automated microfluidic NMR for quantitative protein-ligand interaction analysis
In recent years, there has been a growing trend towards the miniaturization and automation of life science workflows. This trend has led to the development of novel technologies that can automate complex analytical protocols, making them less laborious and more time-efficient. One such technology is microfluidic nuclear magnetic resonance (NMR), which allows for the quantitative analysis of protein-ligand interactions with minimal sample needs and without the need for user intervention. In this paper, the researchers present an automated microfluidic NMR system that uses protein-detected heteronuclear 2D NMR spectroscopy to quantify protein-ligand interactions.
Protein-ligand interactions play a crucial role in many biological processes, and their quantitative characterization is essential for understanding cellular signaling and other life processes. However, the titrations required to measure these interactions can be laborious and time-consuming, often requiring large sample sizes. The microfluidic NMR titration system presented in this paper overcomes these challenges by bringing the analytical power of NMR detection in line with the trend of miniaturization and automation in life science workflows. The system consists of several hardware components whose actions are coordinated by a microcontroller. A transmission line probe, optimized for double-resonance protein detection, accepts planar microfluidic devices, providing considerable freedom in the design of the microfluidic chip. The chip houses six pneumatic on-chip valves, which are used to control the movement of liquid on the chip and a 2.5 μL detection chamber positioned in the volume of maximal probe sensitivity.
“The present work shows how the analytical power of NMR detection can be brought in line with the trend of miniaturization and automation in life science workflows.“, the authors explained.
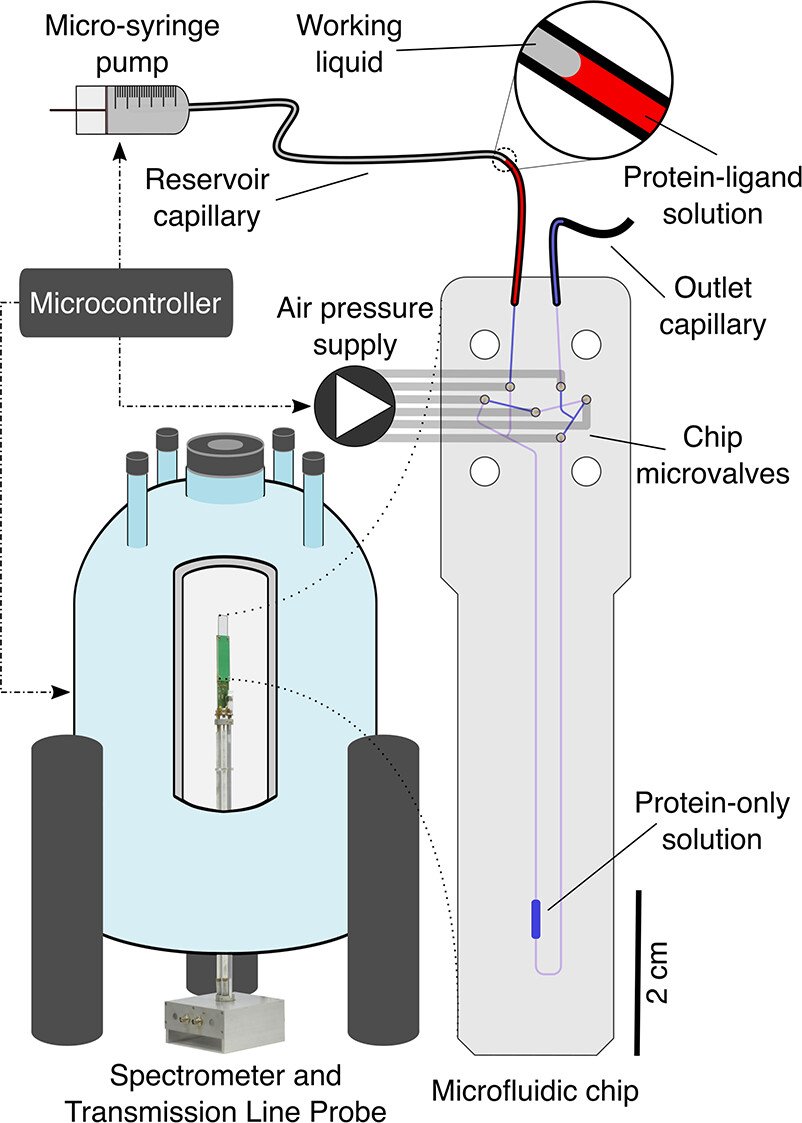
“Schematic representation of the device and its operation inside the NMR spectrometer after the initial filling procedure carried out by the experimenter.” Reproduced from Marek Plata, Manvendra Sharma, Marcel Utz, and Jörn M. Werner Journal of the American Chemical Society 2023 145 (5), 3204-3210 DOI: 10.1021/jacs.2c13052 under Creative Commons Attribution 4.0 International (CC BY 4.0).
In this work, the authors demonstrate the potential of automated microfluidic NMR for quantifying protein-ligand interactions. The microfluidic NMR titration experiment requires a protein-only solution and a solution with the same protein concentration and a molar excess of ligand sufficient to saturate binding. To start the titration experiment, 10 μL of the protein-only solution is injected into the chip using the microsyringe while the middle valve of the bridge pathway is closed. Afterward, a 2 ± 0.25 μL volume of the protein-ligand solution is injected into the mixing circuit, mixed with the previously analyzed solution, and further spectra are acquired. The fully automated process delivers a series of two-dimensional heteronuclear single quantum coherence (2D HSQC) spectra as a function of the ligand to protein ratio, from which the binding process can be quantified.
The results of this study demonstrate the potential of microfluidic NMR for automation of complex analytical protocols, which would otherwise be laborious and time-consuming to perform. This aligns well with a general trend in the life sciences toward miniaturization and automation. The current platform that is working with microliter volumes brings the analytical power of NMR in line with microliter protein protein expression systems for generating fully automated discovery workflows. Further improvements could be realized by combining recent advances in autonomous processing of protein NMR data. The system’s modularity and flexibility of the microfluidic chip design make it easily adaptable to suit specific experimental needs, such as screening applications, protein-protein interactions, and studies of multiligand equilibria. A particularly exciting prospect is the integration of hyperpolarized binding studies in a microfluidic platform. The experiment only uses a modest amount of protein, about 0.7 mg in the current implementation. There is significant scope for further reduction of the total amount of protein and the protein concentration used for the experiment. Efforts in this direction are underway in the researchers’ laboratory and will be reported on at a later occasion.
“In summary, we have successfully automated protein–peptide binding analyses using microfluidic NMR. The results clearly demonstrate the potential of microfluidic NMR for automation of complex analytical protocols, which would otherwise be laborious and time-consuming to perform. This aligns well with a general trend in the life sciences toward miniaturization and automation. “, the authors explained.
Figures and the abstract are reproduced from Marek Plata, Manvendra Sharma, Marcel Utz, and Jörn M. Werner Journal of the American Chemical Society 2023 145 (5), 3204-3210 DOI: 10.1021/jacs.2c13052 under Creative Commons Attribution 4.0 International (CC BY 4.0)
Read the original article: Fully Automated Characterization of Protein–Peptide Binding by Microfluidic 2D NMR


